Don Mercer
We have previously looked the formation of soap scum in water. The culprit was calcium ions that grabbed onto soap molecules and formed an ugly and annoying insoluble compound.
What we must do to prevent soap scum from forming is remove the calcium ions from what we commonly call “hard water”. Fortunately, this is not particularly difficult to accomplish. Many of you are already familiar with the use of water softeners for such a task. However, do you understand how they actually work?
The water softening process takes place on the surface of tiny resin beads which are packed into what is usually a cylindrical column. As we go through our description, you need to keep in mind that all of this is happening at the level of atoms or ions, so none of it is visible to us.
Each plastic-like bead is covered with many active sites to which a sodium ion is attached. Step 1 in the accompanying diagram represents an ion exchange bead and shows only four of these sites. Each site is occupied by a single sodium ion. Step 2 in the diagram brings two calcium ions into the water around the ion exchange resin. Since they have a double positive charge, each calcium ion can force two singly charged sodium ions off the resin bead and take over the vacated active sites as shown in Step 3.
After being replaced by the calcium ions, there will be two sodium ions floating around freely in the water for every calcium ion that was removed by the ion exchange resin bead. With the calcium removed, the ‘soft” water can be used in applications where soap scum would previously have been a problem. Step 4 in the diagram shows the four sodium ions being released into the softened water.
Eventually, a point will be reached where all the sodium ions that were on the beads have been replaced by calcium ions. With its ability to exchange any more sodium ions for calcium ions exhausted, the ion exchanger can no longer soften any more water, and it needs to be regenerated.
The first step in regenerating or recharging the ion exchange column is to shut off the flow of incoming water. Then the system flips to allow a saturated brine or salt solution to be pumped from a reservoir into the column. The resin beads will be allowed to soak for a period of time in the strong salt solution. To avoid disrupting your day-to-day routines, the regeneration is usually done during the night.
The overwhelming concentration of sodium ions in the brine solution will essentially gang up on the calcium ions that are present on the beads and force them off the resin. This is a case where there is strength in numbers, and the calcium ions simply cannot maintain their hold on the active sites of the resin beads.
After regeneration, the active sites on the resin beads will be fully occupied by sodium ions. The solution in the column will contain the displaced calcium ions as well as an excess of sodium ions. By automatically switching a few valves, a short rinse of hard water can be run through the column to rid it of the calcium and sodium solution, which is usually directed to a drain in a sink or in the floor.
After the rinse cycle, the valves are reset allowing hard water to enter the column and softened water to flow through to various locations within the house.
There are a few things to keep in mind when operating a water softener. You don’t want to regenerate your water softener too often because this wastes salt which is a cost factor, and puts salt into the environment. If not done properly, excess salt can be a negative impact on your piping. You should consult a reputable company to optimize the operation of your system to suit your needs.
It should be recognized that while calcium is being taken out of the water, it is being replaced with sodium ions. There may be some potential impact on your body’s sodium levels. To avoid drinking water with elevated sodium levels, it may be advisable to have one tap in your kitchen that delivers unsoftened (i.e., hard) water for you to drink. Once again, it is best to discuss the situation with your water softener supplier.
Hopefully, we have now removed a bit of the mystery surrounding what’s going on inside your water softener.
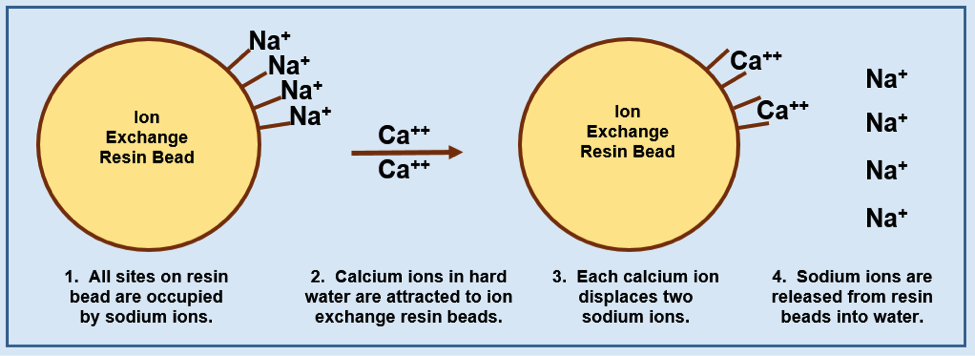
The diagram above shows how sodium ions are released from the ion exchange resin beads and are replaced by calcium ions.
Dr Don Mercer is Associate Professor in Food Science, Department of Food Science, University of Guelph, Guelph, Ontario N1G 2W1, Canada; e-mail: dmercer@uoguelph.ca
Permission to reproduce this article is greatly appreciated and acknowledged.
IUFoST Scientific Information Bulletin (SIB)
FOOD FRAUD PREVENTION